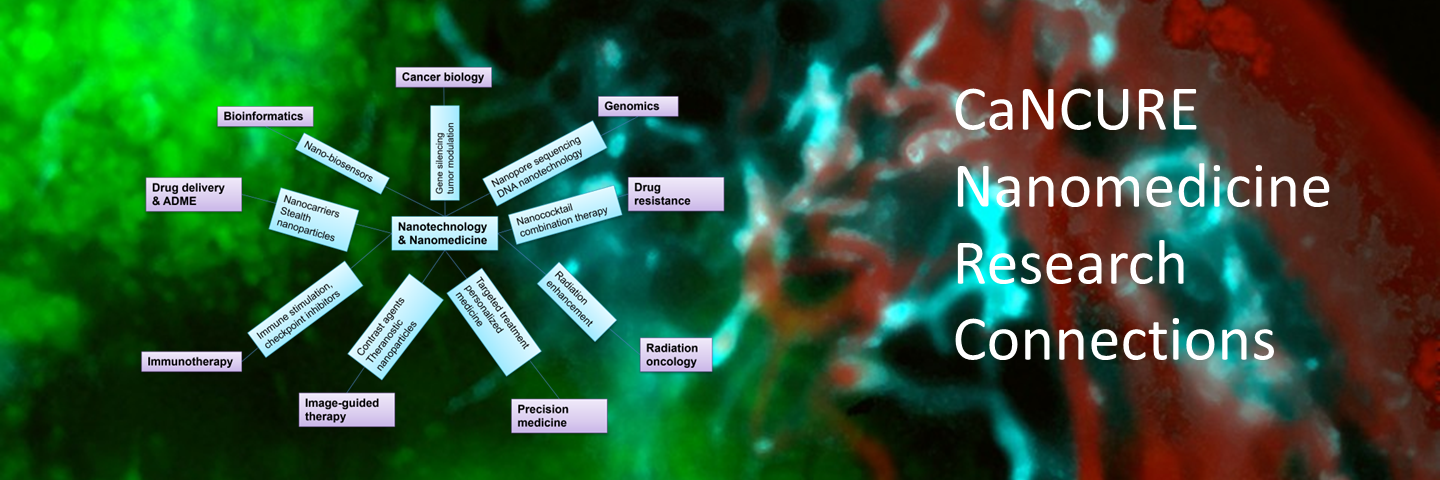
Cancer Nanomedicine Research

Genomic sequencing of translocations in multiple myeloma patients of differing stages
Sustained-release talazoparib for treatment of BRCA1-deficient ovarian cancer
Understanding and overcoming resistance to PARP inhibitors in ovarian cancer patients
Rapid hormone receptor classification using molecular profiling & imaging of breast cancer
MRI imaging of human vascular dysfunction using ferumoxytol nanoparticles
Molecular imaging for detection of gastrointestinal & pancreatic cancers
Resistance to immunogenic cell death in the treatment multiple myeloma with bortezomib
Gadolinium-based nanoparticles for lung cancer radiation dosage enhancement
Genetic differences between early-onset and late-onset colorectal cancer patients
Improving anti-VEGF treatments through exercise training
Nanoformulated PARP inhibitors for treatment of ovarian & breast cancers
Paclitaxel polymer films to prevent local recurrence of sarcoma in murine models
Granzyme M-/Gasdermin E-induced pyroptosis conservation in mice
Role of TP53 in persister cell formation and chemotherapy resistance
Role of RPS15 mutations on chronic lymphocytic leukemia development
Evaluation of implantable anti-cancer therapeutics for localized drug delivery
Focused ultrasound for blood brain barrier disruption and tumor ablation
miR-710 as a therapeutic target in human metastatic breast cancer
Drug/Metabolite quantification of cancer tissue samples using MALDI-MS
Sustained release of nanoTalazoparib for ovarian cancer treatment
Surface-enhanced spatially offset resonance raman spectroscopy for cancer detection
Nano-smart radiotherapy biomaterials for combining radiotherapy & immunotherapy
Defining the t(4,14) mechanism in multiple myeloma progression
Lymphatic drainage and immunosurveillance of brain tumors
Extracellular vesicle characterization for early detection of paclitaxel resistance in triple negative breast cancer
Tumor treatment by intra- or peri-tumoral injection of [177Lu]-Feraheme nanoparticles
Combining radiolabeled drugs & PET to study cancer cell-mediated immune response
Flavonoid-loaded ACE2 nanodrones to target lung cancer cells
Ferumoxytol for non-invasive magnetic resonance imaging of neoplastic microvasculature
Development & evaluation of a theranostic probe for image-guided treatment of oral cancer
Leveraging the synergistic anti-proliferative effect of SHMT2 and CDK4 in triple negative breast cancer
Silica-based bismuth gadolinium nanoparticles for radiation dose enhancement in lung cancer
The research experience doesn't stop with the co-op
Over half our trainees choose to continue their CaNCURE research after their co-op. CaNCURE trainees have advanced their research projects while earning credit toward Senior Capstone, Directed Study, and Honors Senior Thesis courses. OurCaNCURE staff is available to help!
Opportunities for Further Education & Training
Looking for more ways to gain real-world skills and experience in cancer nanomedicine? We offer a variety of experiential learning courses that are open to all CaNCURE alumni. Come put the ideas you are learning into action!
Nanomedicine
Learn & blog about enabling innovations in nanomedicine (NNMD 5270)
Commercialization
Create a virtual start-up company under industry mentorship (NNMD 5470)
Directed Study
Earn credit for continuing CaNCURE research (NNMD 4991)
Research Techniques
Conduct experiments in a live hands-on & online laboratory (NNMD 5370)
Seminars & Workshops
Engage with research, clinical, and industry experts (NNMD 5272 & 5274)




