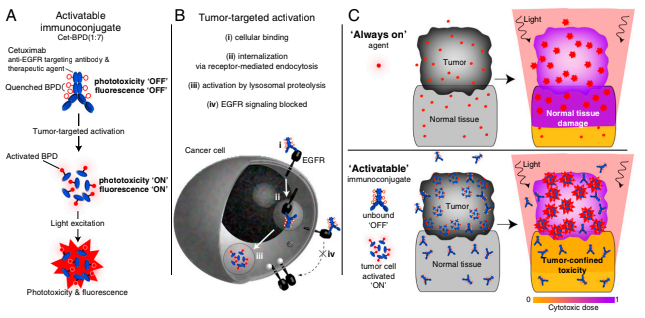
Oral squamous cell carcinoma frequently exhibits overexpression of the epidermal growth factor receptor (EGFR), presenting an opportunity for targeted therapeutic and imaging strategies. Current surgical practices rely primarily on white-light visualization, often removing an additional margin of surrounding tissue, yet residual microscopic tumor cells may remain undetected. My research project explores the use of a dual-function, activatable EGFR-targeted immunoconjugate designed to enable both tumor-specific fluorescence imaging and photodynamic therapy (PDT). Initial in vitro studies will analyze immunoconjugate binding and activation in EGFR-expressing cells using fluorescence-activated cell sorting. These results will then be extended to in vivo mouse models to investigate the utility of the immunoconjugate for fluorescence-guided tumor visualization and PDT-mediated tumor destruction. PDT will be explored both before and after surgical resection to evaluate its potential role in eliminating residual tumor cells. By comparing imaging-guided and non-imaging-guided surgical approaches, as well as pre- and post-resection imaging, this work aims to evaluate whether activatable photoimmunotherapy can improve surgical precision and reduce residual tumor burden. Ultimately, this project seeks to advance a targeted, image-guided therapeutic strategy for oral cancer.