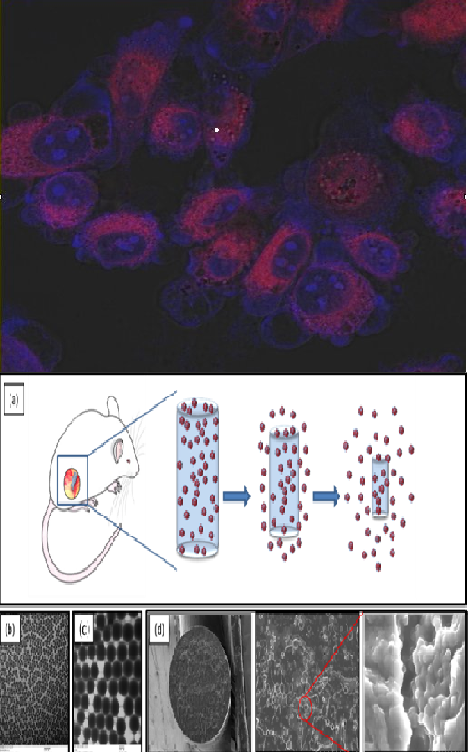
Prostate cancer is the second leading cause of cancer-related deaths in men. While 5-year survival rates are high, prostate cancer is slow-growing and recurring tumors are often unresponsive to treatment. Salvage brachytherapy offers a second chance for cure but leads to high rates of rectal toxicity. The Sridhar lab, in collaboration with radiation oncologists at Dana Farber Cancer Institute, aims to locally radiosensitize the prostate throughout the duration of brachytherapy treatment to enable the use of lower radiation doses with lower toxicities for a greater chance of cure. Currently, brachytherapy uses inert spacers for spatial guidance during the one-time injection of radioactive seeds. We leverage the use of these spacers to create a new modality of chemo-radiation therapy, replacing the spacers routinely used in brachytherapy with our own biodegradable spacers doped with radiosensitizing drug Docetaxel, both with and without silica nanoparticles. Here the silica nanoparticles would allow for a highly controlled dual release platform in which first the nanoparticle is released from the spacer and second the drug is released from the nanoparticle. This approach provides localized in-situ delivery of the chemotherapeutic sensitizer directly to the tumor with enhanced uptake and avoids the toxicity associated with current systemic delivery. Complete understanding and characterization of the release kinetics of both the spacer and nanoparticle is crucial in developing an effective slow-sustained release of local chemotherapy with minimal adverse toxicities. These biodegradable implants have a robust platform and can incorporate a variety of different chemotherapeutics, nanoparticles, or biologics as a local delivery modality for varying diseases. As part of the CaNCURE project, we will continue to complete in vitro studies needed to understand the underlying properties and release of the spacer, the therapeutic efficacy of nanoparticles versus free chemotherapeutics, and the cellular uptake of the drug and particles. We will also continue to monitor the therapeutic impact in vivo of Docetaxel loaded spacers in PC3 xenografted mice. A full toxicity study will be completed to ensure the safety of the new device in comparison to currently used taxotere, in hopes of developing a safe and translatable new modality of chemotherapy.